Lupus Discovery
Lupus Discovery

Accelerated
Turnaround Time

Cost-
Efficiency

Modular
Platform

Tailored
Study Design
High patient variability and multi-organ
involvement (skin, kidney, CNS) complicate
clinical trial designs.
Despite advances, long-term remission
remains elusive, demanding novel
MOA-based therapies.
Inadequate scientific rigor in reproducibility
and precision directly escalates
the risk of late-stage clinical attrition.

 Target-Specific Model Selection:
Guidance on choosing MRL/Faslpr vs NZW/B F1 based on your MOA.
Target-Specific Model Selection:
Guidance on choosing MRL/Faslpr vs NZW/B F1 based on your MOA.
 Customized Endpoints:
Beyond survival—incorporating diverse biomarkers & pathology scoring.
Customized Endpoints:
Beyond survival—incorporating diverse biomarkers & pathology scoring.
 Bridging Data Gaps:
Correlating preclinical efficacy with potential clinical outcomes.
Bridging Data Gaps:
Correlating preclinical efficacy with potential clinical outcomes.
 Go/No-Go Decision Support:
Objective data analysis to de-risk your pipeline before IND.
Go/No-Go Decision Support:
Objective data analysis to de-risk your pipeline before IND.
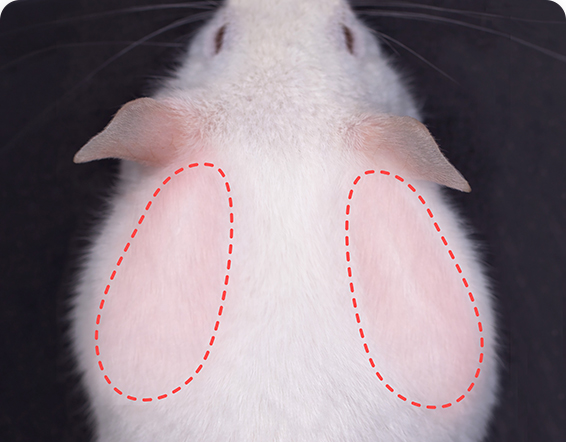
 Rapid Efficacy Screening:
Rapid Efficacy Screening:
Aggressive T cell driven
onset enabling accelerated
Go/No-Go decision making.
 Multi-organ involvement:
Multi-organ involvement:
including massive
lymphadenopathy and
cutaneous lesions.
 Chronic Disease
Chronic Disease
Evaluation:Gold standard
for assessing long-term
survival and chronic lupus
nephritis progression.
 Clinically relevant renal
Clinically relevant renal
failure: Driven by chronic
autoantibody-mediated
immune complex deposition.
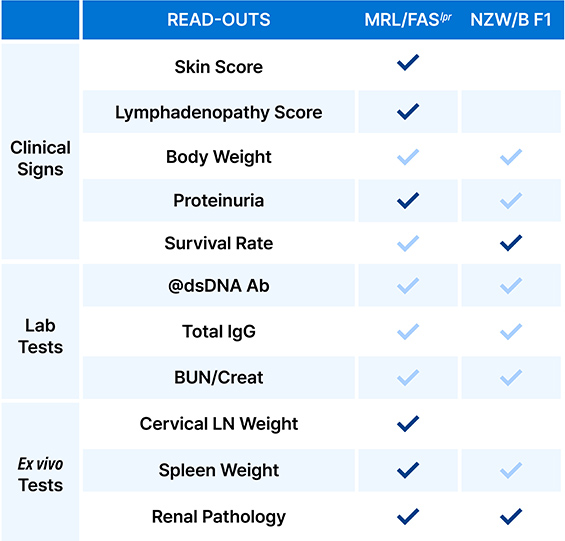
We offer distinct murine models optimized for your
candidate’s specific immunological target and study timeline.
 Clinical: Body weight, Skin score, Proteinuria
Clinical: Body weight, Skin score, Proteinuria
 Immunological: Autoantibody (anti-dsDNA), FACS analysis
Immunological: Autoantibody (anti-dsDNA), FACS analysis
 Pathological: Glomerulonephritis & Interstitial scoring
Pathological: Glomerulonephritis & Interstitial scoring
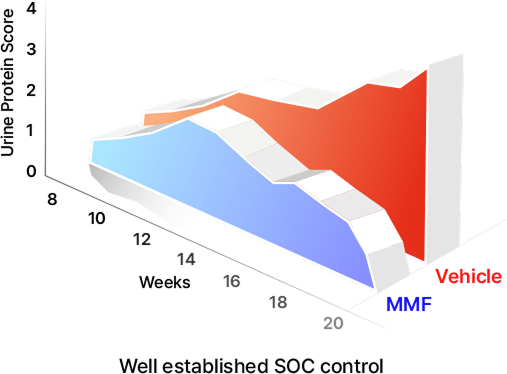
 Integrated Endpoints : Clinical and pathological measures
Integrated Endpoints : Clinical and pathological measures
capturing both renal function and tissue-level disease changes,
serving as the primary indicators of therapeutic efficacy
 Key Functional Readout :Quantitative assessment of
Key Functional Readout :Quantitative assessment of
roteinuria supported by renal histopathological evaluation
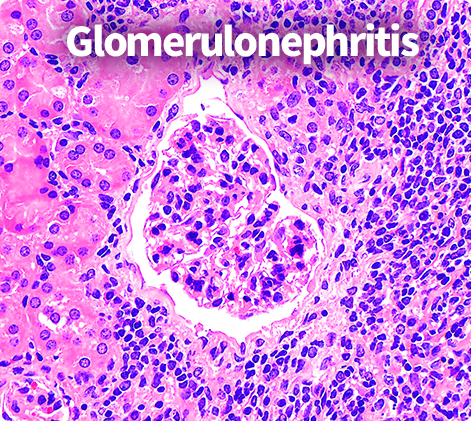
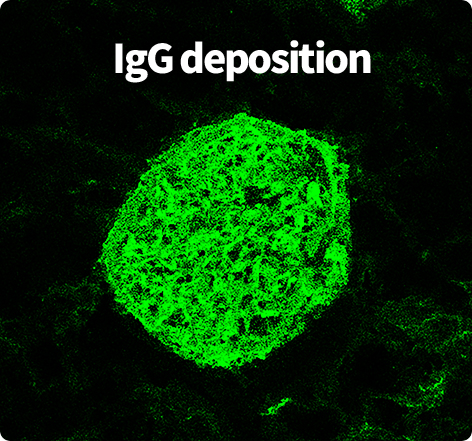

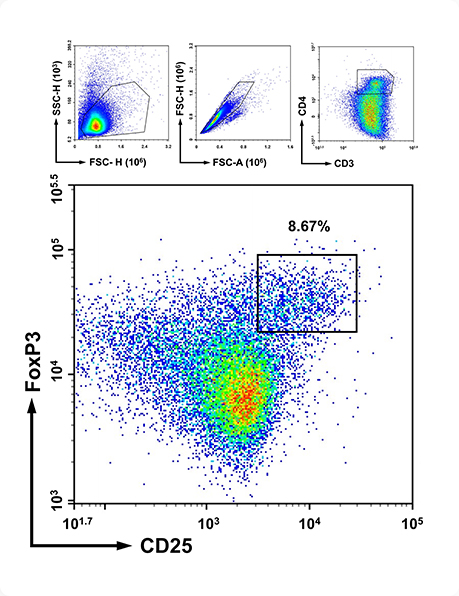
Through in-depth analysis of anti-dsDNA antibodies, C4 concentrations,
and flow cytometry, Preclina enables detailed tracking of therapeutic response
and data-driven development decisions.
Unlock validated analytical insights with Preclina and drive your project forward
with precision.