|
|
|
||||||||||||||||
Strategic Lupus Model Portfolio |
||||||
|
Aligning Drug MOA with Disease Pathogenesis |
||||||
|
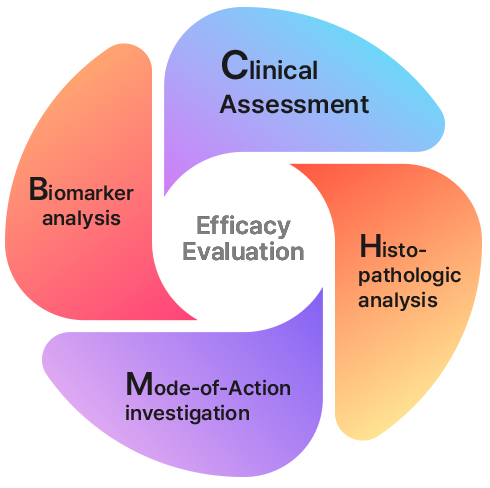
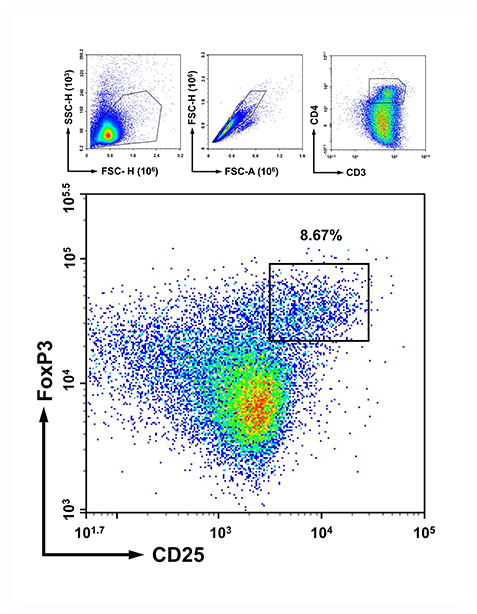
Multidimensional Profiling |
||||
|
From Clinical Phenotypes to Molecular Mechanisms |
||||
|
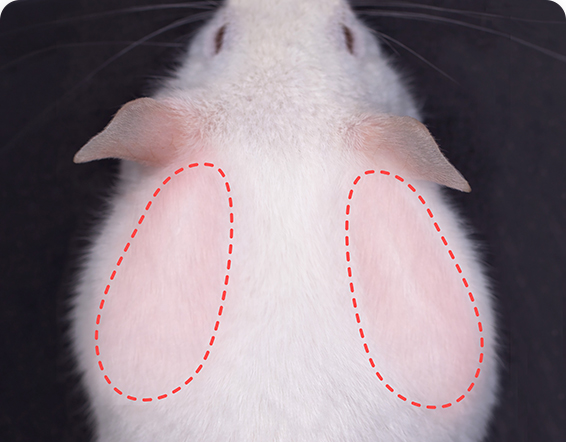
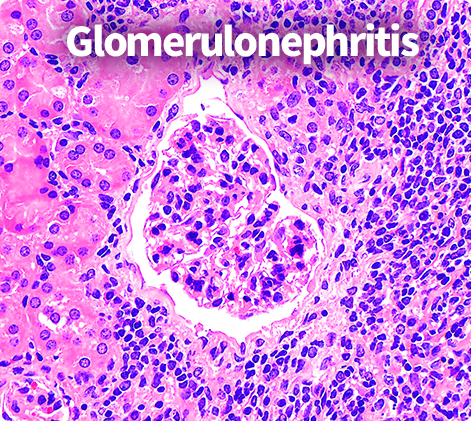
Precision Renal Pathology &
|
||
|
Mechanistic Insight &
|
||||||||
|
||||||||
|
Strategic Partnership for
|
|
Accelerating Discovery with an Integrated PreVIVO™ Platform |
|

|
Follow us
|
|||||
|
[ HO ] 2206, Songdo AIT Center, Yeonsu-gu, Incheon, South Korea [ HO ] 719, Teratower B, Songpa-daero, Songpa-gu, Seoul [ BO ] 203, 1st Bldg, Chilgokjungang-daero 136-gil, Buk-gu, Daegu [ Preclina Europe ] Hegenheimermattweg 167A, Basel, Switzerland [ Preclina US ] CIC Cambridge, One Broadway, Boston, MA, USA |